
Structural Basis for dsRNA Recognition, Filament Formation, and Antiviral Signal Activation by MDA5: Cell

RNF122 suppresses antiviral type I interferon production by targeting RIG-I CARDs to mediate RIG-I degradation | PNAS

Elimination of negative feedback in TLR signalling allows rapid and hypersensitive detection of microbial contaminants | Scientific Reports

Structural insights on dsRNA-mediated activation of RIG-I. RIG-I is an... | Download Scientific Diagram
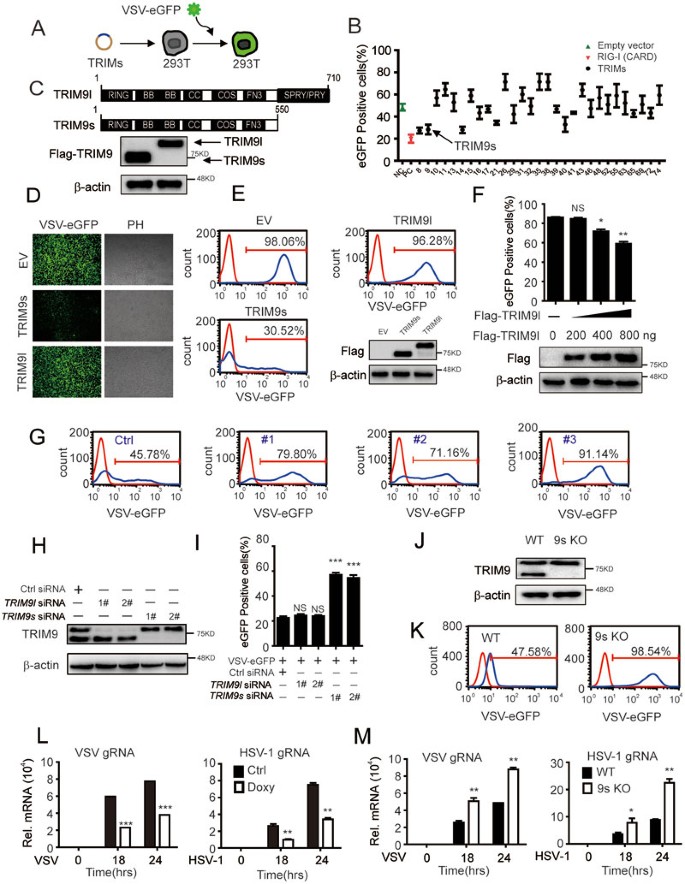
TRIM9 short isoform preferentially promotes DNA and RNA virus-induced production of type I interferon by recruiting GSK3β to TBK1 | Cell Research

RNF122 suppresses antiviral type I interferon production by targeting RIG-I CARDs to mediate RIG-I degradation | PNAS
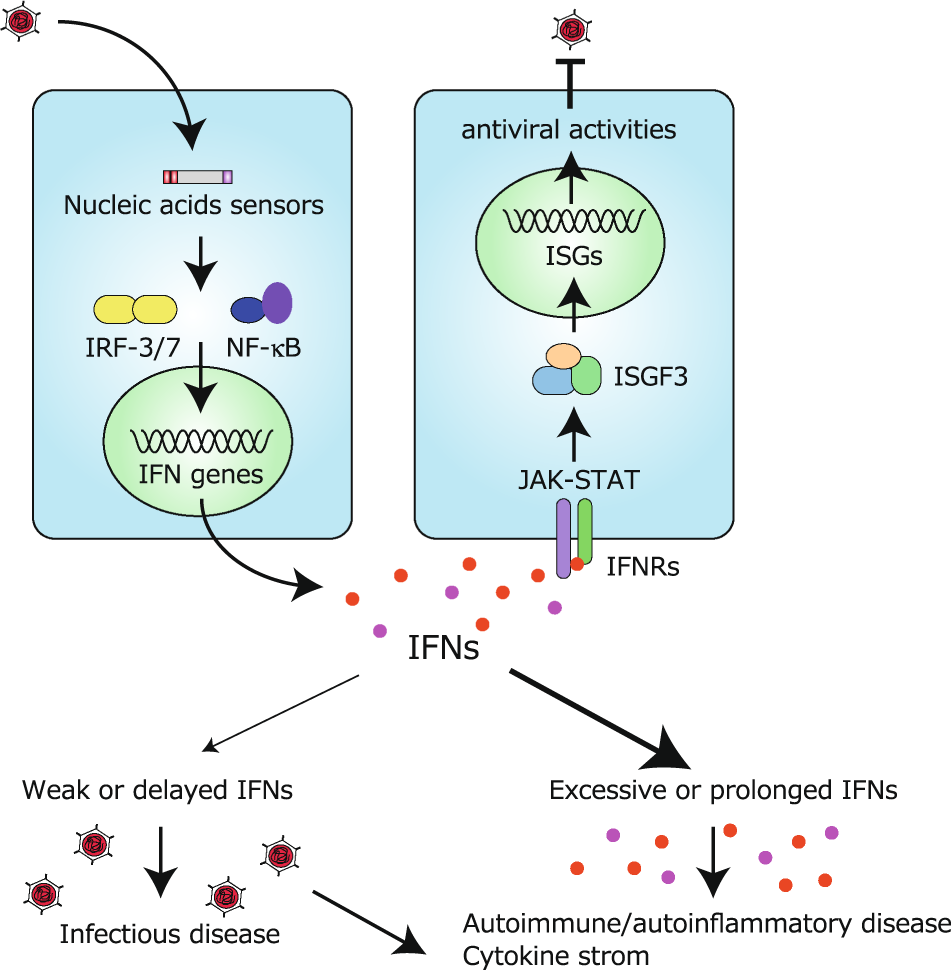
Regulation of RIG-I-like receptor-mediated signaling: interaction between host and viral factors | Cellular & Molecular Immunology

Attenuation of Antiviral Immune Response Caused by Perturbation of TRIM25-Mediated RIG-I Activation under Simulated Microgravity - ScienceDirect
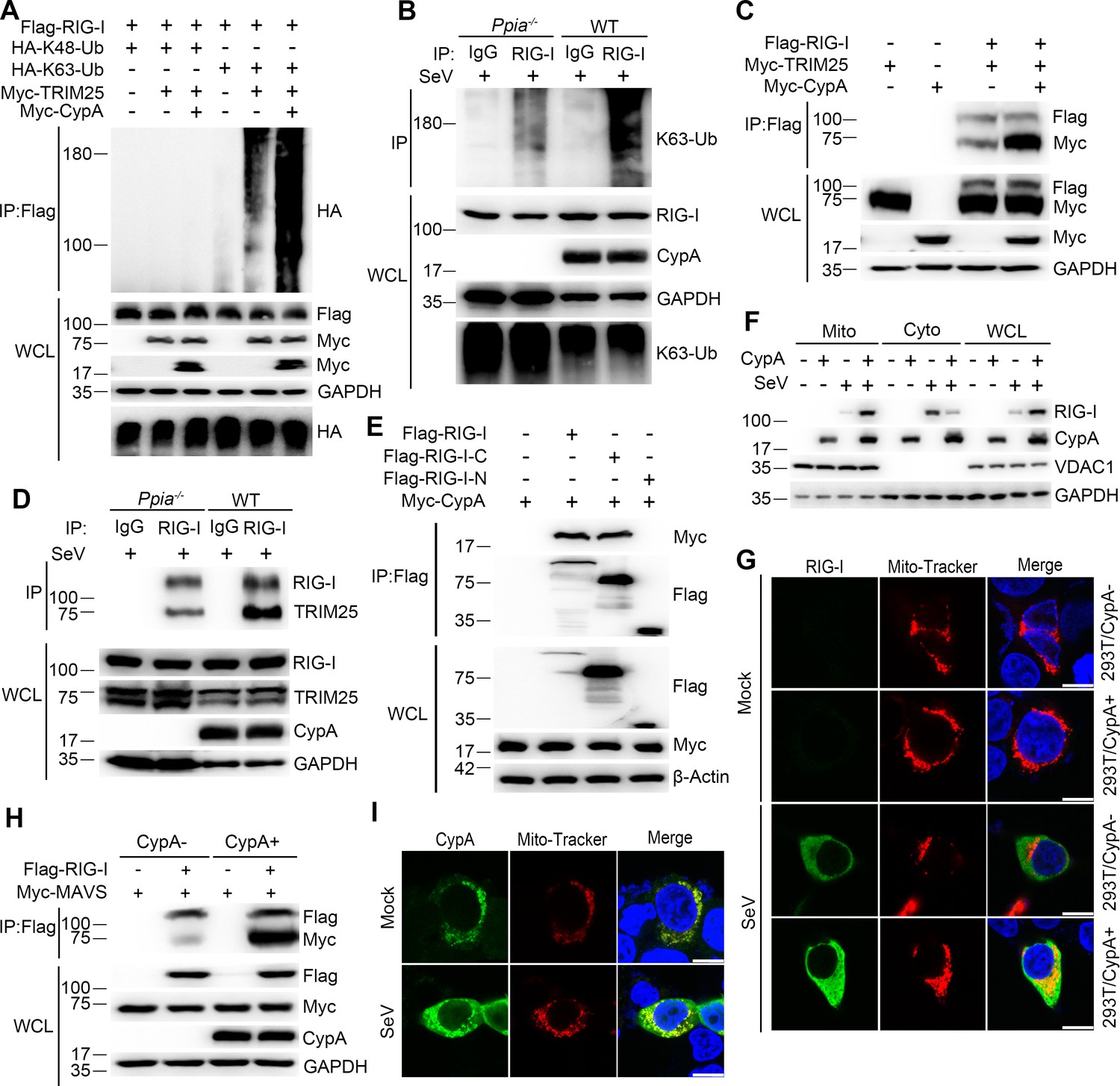
Cyclophilin A-regulated ubiquitination is critical for RIG-I-mediated antiviral immune responses | eLife

The C-Terminal Domain of Salmonid Alphavirus Nonstructural Protein 2 (nsP2) Is Essential and Sufficient To Block RIG-I Pathway Induction and Interferon-Mediated Antiviral Response | Journal of Virology

Foot-and-Mouth Disease Virus 3B Protein Interacts with Pattern Recognition Receptor RIG-I to Block RIG-I–Mediated Immune Signaling and Inhibit Host Antiviral Response | The Journal of Immunology

RIG-I Forms Signaling-Competent Filaments in an ATP-Dependent, Ubiquitin-Independent Manner - ScienceDirect





![Recombinant Anti-RIG-I/DDX58 antibody [EPR18629] KO Tested (ab180675) | Abcam Recombinant Anti-RIG-I/DDX58 antibody [EPR18629] KO Tested (ab180675) | Abcam](https://www.abcam.com/ps/products/180/ab180675/Images/ab180675-250348-anti-rig-i-ddx58-antibody-epr18629-immunoprecipitation.jpg)
![Recombinant Anti-RIG-I/DDX58 antibody [EPR18629] KO Tested (ab180675) | Abcam Recombinant Anti-RIG-I/DDX58 antibody [EPR18629] KO Tested (ab180675) | Abcam](https://www.abcam.com/ps/products/180/ab180675/Images/ab180675-450433-anti-rig-iddx58-antibody-epr18629-western-blot-wildtype-a549-ddx58-knockout-a5492.jpg)


![Recombinant Anti-RIG-I/DDX58 antibody [EPR18629] KO Tested (ab180675) | Abcam Recombinant Anti-RIG-I/DDX58 antibody [EPR18629] KO Tested (ab180675) | Abcam](https://www.abcam.com/ps/products/180/ab180675/Images/ab180675-250351-anti-rig-i-ddx58-antibody-epr18629-western-blot.jpg)



